Case Number: 25MST023
Manager: Robert Prosak
Licensing Associate, Business Development
S&T Technology Transfer & Economic Development
robert.prosak@mst.edu
PDF Download: NeuProIO Marketing Slick
Image Download: NeuProIO.jpg
Journal Publication Download: NeuProIO Publication
Opportunity
Seeking a licensing and development partner to advance the program to clinical studies and onward towards commercialization
Problem Statement
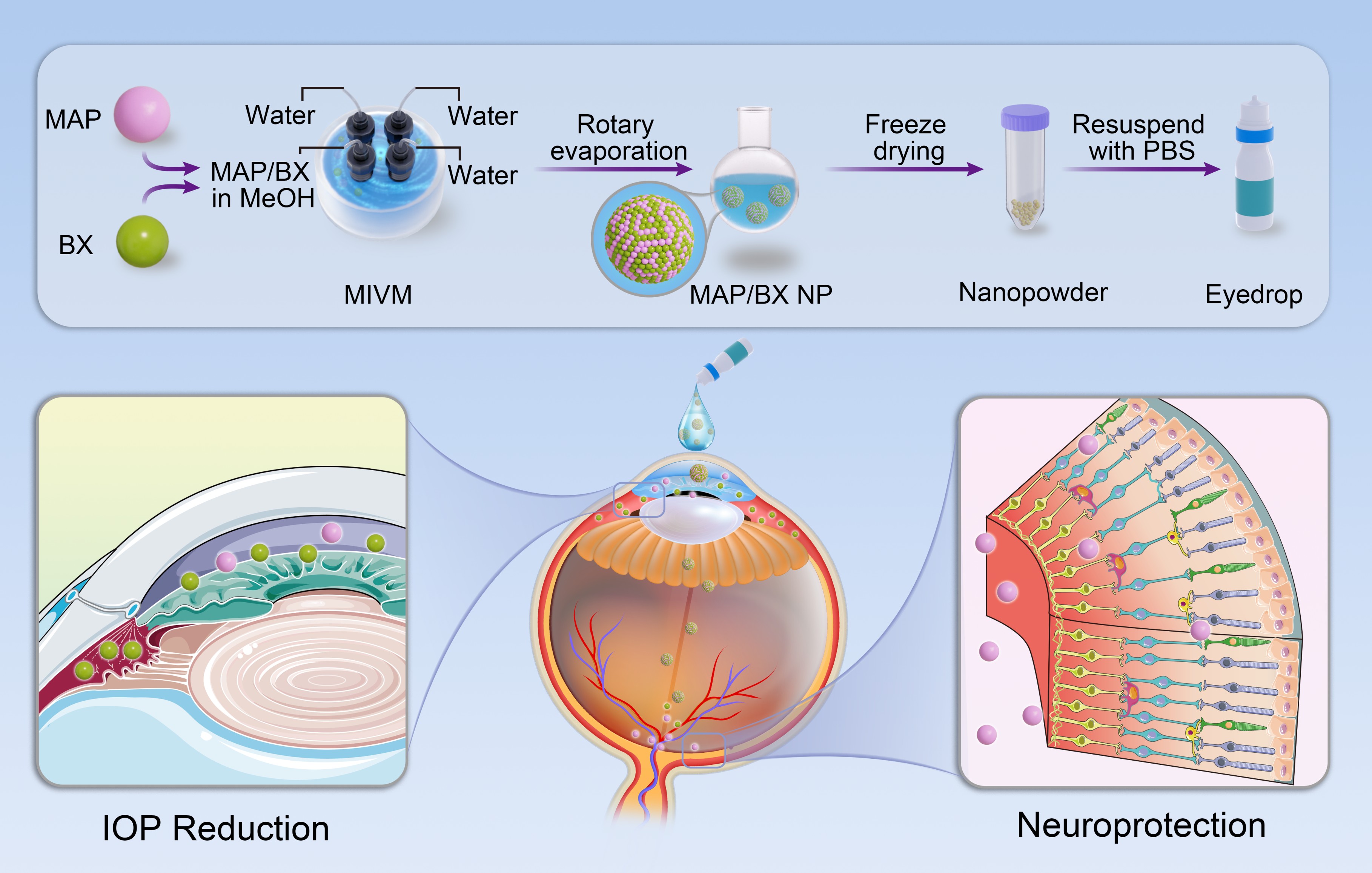
Glaucoma is the leading cause of blindness worldwide with around 80 million affected worldwide. For those suffering from glaucoma, using eyedrops is the least invasive method for symptomatic relief. Current eyedrop products are solely targeting intraocular pressure (IOP) reduction. However, IOP reduction alone is insufficient to halt disease progression. A significant proportion of glaucoma patients still experience progressive damage to retinal ganglion cell (RGC) and the optic nerve even following IOP-lowering treatments. There is an unmet need for novel ocular drug formulations that can simultaneously target IOP reduction and neuroprotection.
Solution
Researchers at Missouri University of Science and Technology and Stanford University have developed a new solid drug nanoparticle (NeuProIO) with two active agents, a neuroprotectant maprotiline and a beta-adrenergic blocker betaxolol. NeuProIO has shown improved biosafety, ocular biocompatibility, tissue permeability, sustained intraocular pressure (IOP) lowering and neuroprotective effect. More importantly, betaxolol in NeuProIO synergistically improved the delivery of the maprotiline to the posterior eye after topical administration, thereby enhancing its neuroprotective effect.
Value Proposition
NeuProIO emphasizes scalable manufacturing, ease of administration, improved ocular biocompatibility, enhanced bioavailability, and simultaneous IOP reduction and neuroprotection. This innovation not only sheds new light on the potential of combination therapy in glaucoma treatment but also presents a scalable solution for the industrial production of advanced ocular drug formulations, ultimately improving patient outcomes and compliance.
Development Stage
Demonstrated efficacy in vivo glaucoma animal models for IOP reduction and neuroprotection studies.
Patent Status
Provisional Patent Application Filed
Publication
https://doi.org/10.1016/j.cej.2026.173145
Investors
Lei Xu, PhD candidate, Dong Liu, MD, PhD, Yang Hu, MD, PhD, and Hu Yang, PhD

Follow Technology Transfer and Economic Development